Atoms Combine in Whole Number Ratios!
Chemical formulas and equations are based on the Law of Definite Proportions. Video by Khan Academy.
Strategies for Success:
- Coefficients are used for balancing equations. They are the number in front of compounds or elements.
- Subscripts come after the element symbol and indicate the number of that element. In H2O the "2" means there are two H atoms.
- If there is no subscript after an element symbol, we assume it to be "1". In H2O this means there is "1" O atom.
Key Information
When balancing chemical equations our goal is to have the same number of each type of atom on both sides of the equation.
You can only change the coefficients when balancing!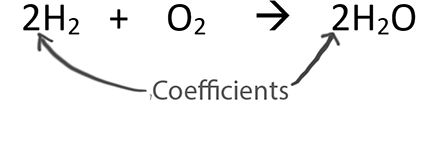
Balanced equations provide ratios between substances. The coefficients can be thought of as mole ratios.